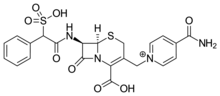
Cefsulodin
 |
|
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Identifiers | |
|
|
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| ECHA InfoCard | 100.052.431 |
| Chemical and physical data | |
| Formula | C22H21N4O8S2+ |
| Molar mass | 533.556 g/mol |
| 3D model (Jmol) | |
|
|
|
|
|
|
|
Cefsulodin is a third-generation cephalosporin antibiotic with specific activity against Pseudomonas aeruginosa. It has no significant activity against other Gram-negative bacteria and very limited activity against Gram-positive bacteria and anaerobic bacteria. Cefsulodin was first synthesized and patented by the Takeda Pharmaceutical Company in 1977. In 2002, Takeda stopped production of cefsulodin. Many years of low-stability cefsulodin production has led to a widespread reduction of laboratory and research uses. Current attempts (i.e. IDEXX Laboratories) of increasing purity and stability of cefsulodin center around recrystallization. Typically, the process entails: Cefsulodin is dissolved in an organic solvent, sodium ions, water, or any mixture thereof, then subsequently recrystallized through separation of the unwanted fraction. Recently, TOKU-E has found the main cause of cefsulodin instability stems from one key impurity in 7-aminocephalosporanic acid, a raw material used in the synthesis of cefsulodin. To produce high-purity, high-stability cefsulodin, TOKU-E uses industrial HPLC to remove significant quantities of this impurity in 7-ACA and thus produces ultrapure, ultrastable, and ultrapotent cefsulodin.
Cefuslodin is most commonly used in cefsulodin-irgasan-novobiocin agar to select for Yersinia microorganisms. This agar is most often used in water and beverage testing.
The following represents MIC susceptibility data for various P. aeruginosa strains.
...
Wikipedia
