Squalene
 |
|
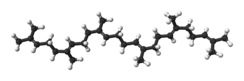 |
|
| Names | |
|---|---|
|
Systematic IUPAC name
(6E,10E,14E,18E)-2,6,10,15,19,23-Hexamethyltetracosa-2,6,10,14,18,22-hexaene
|
|
| Identifiers | |
|
111-02-4 |
|
| 3D model (Jmol) | Interactive image |
| 3DMet | B00166 |
| 1728919 | |
| ChEBI |
CHEBI:15440 |
| ChEMBL |
ChEMBL458402 |
| ChemSpider |
553635 |
| ECHA InfoCard | 100.003.479 |
| EC Number | 203-826-1 |
| 3054 | |
| KEGG |
C00751 |
| MeSH | Squalene |
| PubChem | 638072 |
| RTECS number | XB6010000 |
| UNII |
7QWM220FJH |
|
|
|
|
| Properties | |
| C30H50 | |
| Molar mass | 410.73 g·mol−1 |
| Appearance | Pale yellow, translucent liquid |
| Density | 0.858 g cm−3 |
| Melting point | −5 °C (23 °F; 268 K) |
| Boiling point | 285 °C (545 °F; 558 K) at 3.3 kPa |
| log P | 12.188 |
| Viscosity | 12 cP (at 20 °C) |
| Hazards | |
| NFPA 704 | |
| Flash point | 110 °C (230 °F; 383 K) |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
|
|
|
| Infobox references | |
Squalene is a natural 30-carbon organic compound originally obtained for commercial purposes primarily from shark liver oil (hence its name, as Squalus is a genus of sharks), although plant sources (primarily vegetable oils) are now used as well, including amaranth seed, rice bran, wheat germ, and olives. Yeast cells have been genetically engineered to produce commercially useful quantities of "synthetic" squalene.
All plants and animals produce squalene as a biochemical intermediate, including humans. It occurs in high concentrations in the stomach oil of birds in the order Procellariiformes.
Squalene is a hydrocarbon and a triterpene, and is a natural and vital part of the synthesis of all plant and animal sterols, including cholesterol, steroid hormones, and vitamin D in the human body.
Squalene is the biochemical precursor to the whole family of steroids. Oxidation (via squalene monooxygenase) of one of the terminal double bonds of squalene yields 2,3-squalene oxide, which undergoes enzyme-catalyzed cyclization to afford lanosterol, which is then elaborated into cholesterol and other steroids.
Squalene is an ancient molecule. In plants, squalene is the precursor to stigmasterol. In certain fungi, it is the precursor to ergosterol. However, blue-green algae and some bacteria do not manufacture squalene, and must acquire it from the environment if they need it.
...
Wikipedia


